Lipids and Membranes in Pathological Biochemistry


This article, written by Moshtaghi Kashanian of Kerman University of Medical Sciences, explores the role of lipids in biochemistry and their relationship to cell membranes.
- Uploaded on | 6 Views
-
 fevrosiya
fevrosiya
About Lipids and Membranes in Pathological Biochemistry
PowerPoint presentation about 'Lipids and Membranes in Pathological Biochemistry'. This presentation describes the topic on This article, written by Moshtaghi Kashanian of Kerman University of Medical Sciences, explores the role of lipids in biochemistry and their relationship to cell membranes.. The key topics included in this slideshow are . Download this presentation absolutely free.
Presentation Transcript
Slide1Lipids and Membranes Lipids and Membranes Moshtaghi-Kashanian Moshtaghi-Kashanian Pathological Biochemist Pathological Biochemist Kerman University of Medical Sciences Kerman University of Medical Sciences
Slide2LipidsLipids • Lipids: a heterogeneous class of naturally occurring organic compounds classified together on the basis of common solubility properties • Lipids: a heterogeneous class of naturally occurring organic compounds classified together on the basis of common solubility properties • They are insoluble in water, but soluble in aprotic organic solvents, including diethyl ether, chloroform, methylene chloride, and acetone • They are insoluble in water, but soluble in aprotic organic solvents, including diethyl ether, chloroform, methylene chloride, and acetone • Lipids include • Lipids include • triacylglycerols, phosphodiacylglycerols, sphingolipids, glycolipids, lipid-soluble vitamins, and prostaglandins • triacylglycerols, phosphodiacylglycerols, sphingolipids, glycolipids, lipid-soluble vitamins, and prostaglandins • cholesterol, steroid hormones, and bile acids • cholesterol, steroid hormones, and bile acids
Slide3Fatty Acids Fatty Acids • Fatty acid: a long, un-branched chain carboxylic acid, most commonly of 12 - 20 carbons, derived from hydrolysis of animal fats, vegetable oils, or phosphodiacylglycerols of biological membranes • Fatty acid: a long, un-branched chain carboxylic acid, most commonly of 12 - 20 carbons, derived from hydrolysis of animal fats, vegetable oils, or phosphodiacylglycerols of biological membranes • In the shorthand notation for fatty acids • In the shorthand notation for fatty acids • the number of carbons and the number of double bonds in the chain are shown by two numbers, separated by a colon • the number of carbons and the number of double bonds in the chain are shown by two numbers, separated by a colon
Slide4Fatty Acids Fatty Acids
Slide5Fatty Acids Fatty Acids Among the fatty acids most abundant in plants and animals Among the fatty acids most abundant in plants and animals • Nearly all have an even number of carbon atoms, most between 12 and 20, in an un-branched chain • Nearly all have an even number of carbon atoms, most between 12 and 20, in an un-branched chain • The three most abundant are palmitic (16:0), stearic acid (18:0), and oleic acid (18:1) • The three most abundant are palmitic (16:0), stearic acid (18:0), and oleic acid (18:1) • In most unsaturated fatty acids, the cis isomer predominates; the trans isomer is rare • In most unsaturated fatty acids, the cis isomer predominates; the trans isomer is rare • unsaturated fatty acids have lower melting points than their saturated counterparts; the greater the degree of unsaturation, the lower the melting point • unsaturated fatty acids have lower melting points than their saturated counterparts; the greater the degree of unsaturation, the lower the melting point
Slide6TriacylglycerolsTriacylglycerols • Triacylglycerol (triglyceride): an ester of glycerol with three fatty acids • Triacylglycerol (triglyceride): an ester of glycerol with three fatty acids • natural soaps are prepared by boiling triglycerides (animal fats or vegetable oils) with NaOH, in a reaction called saponification (Latin, sapo , soap • natural soaps are prepared by boiling triglycerides (animal fats or vegetable oils) with NaOH, in a reaction called saponification (Latin, sapo , soap
Slide8SoapsSoaps • Soaps form water-insoluble salts when used in water containing Ca(II), Mg(II), and Fe(III) ions (hard water) • Soaps form water-insoluble salts when used in water containing Ca(II), Mg(II), and Fe(III) ions (hard water)
Slide9PhosphoacylglycerolsPhosphoacylglycerols • Phosphoacylglycerols (phosphoglycerides) are the second most abundant group of naturally occurring lipids • Phosphoacylglycerols (phosphoglycerides) are the second most abundant group of naturally occurring lipids • found almost exclusively in plant and animal membranes, which typically consist of 40% -50% phosphoacylglycerols and 50% - 60% proteins • found almost exclusively in plant and animal membranes, which typically consist of 40% -50% phosphoacylglycerols and 50% - 60% proteins • the most abundant phosphoacylglycerols are derived from phosphatidic acid, a molecule in which glycerol is esterified with two molecules of fatty acid and one of phosphoric acid • the most abundant phosphoacylglycerols are derived from phosphatidic acid, a molecule in which glycerol is esterified with two molecules of fatty acid and one of phosphoric acid • the three most abundant fatty acids in phosphatidic acids are palmitic acid (16:0), stearic acid (18:0), and oleic acid (18:1) • the three most abundant fatty acids in phosphatidic acids are palmitic acid (16:0), stearic acid (18:0), and oleic acid (18:1)
Slide10PhosphoacylglycerolsPhosphoacylglycerols • A phosphatidic acid • A phosphatidic acid • further esterification with a low-molecular weight alcohol gives a phosphoacylglycerol • further esterification with a low-molecular weight alcohol gives a phosphoacylglycerol • among the most common of these low-molecular • among the most common of these low-molecular
Slide11PhosphoacylglycerolsPhosphoacylglycerols
Slide12PhosphoacylglycerolsPhosphoacylglycerols
Slide13•A lecithin • A lecithin Phosphoacylglycerols Phosphoacylglycerols
Slide14WaxeWaxe • Esters of long-chain fatty acids and alcohols • Esters of long-chain fatty acids and alcohols • from the Old English word weax = honeycom • from the Old English word weax = honeycom
Slide15SphingolipidsSphingolipids
Slide16GlycolipidsGlycolipids • Glycolipid: a compound in which a carbohydrate is bound to an -OH of the lipid many glycolipids are derived from ceramides • Glycolipid: a compound in which a carbohydrate is bound to an -OH of the lipid many glycolipids are derived from ceramides
Slide17SteroidsSteroids • Steroids: a group of plant and animal lipids that have this tetracyclic ring structure • Steroids: a group of plant and animal lipids that have this tetracyclic ring structure • The features common to the ring system of most naturally occurring steroids are illustrated on the next screen • The features common to the ring system of most naturally occurring steroids are illustrated on the next screen
Slide18SteroidsSteroids
Slide19CholesterolCholesterol
Slide20AndrogensAndrogens • Androgens - male sex hormones • Androgens - male sex hormones • synthesized in the testes • synthesized in the testes • responsible for the development of male secondary sex characteristics • responsible for the development of male secondary sex characteristics
Slide21AndrogensAndrogens
Slide22EstrogensEstrogens • Estrogens - female sex hormones • Estrogens - female sex hormones • synthesized in the ovaries • synthesized in the ovaries • responsible for the development of female secondary sex characteristics and control of the menstrual cycle • responsible for the development of female secondary sex characteristics and control of the menstrual cycle
Slide23EstrogensEstrogens
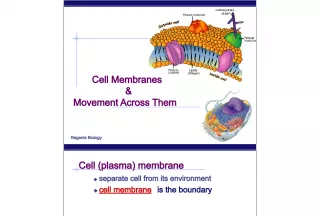
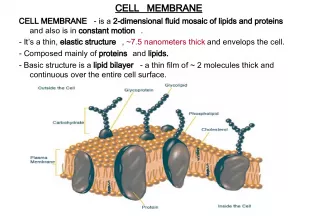
Slide24Biological Membranes Biological Membranes • In aqueous solution, phosphoglycerides spontaneously form into a lipid bilayer, with a back- to-back arrangement of lipid monolayers • In aqueous solution, phosphoglycerides spontaneously form into a lipid bilayer, with a back- to-back arrangement of lipid monolayers • polar head are in contact with the aqueous environment • polar head are in contact with the aqueous environment • nonpolar tails are buried within the bilayer • nonpolar tails are buried within the bilayer • the major force driving the formation of lipid bilayers is hydrophobic interaction • the major force driving the formation of lipid bilayers is hydrophobic interaction • the arrangement of hydrocarbon tails in the interior can be rigid (if rich in saturated fatty acids) or fluid (if rich in unsaturated fatty acids) • the arrangement of hydrocarbon tails in the interior can be rigid (if rich in saturated fatty acids) or fluid (if rich in unsaturated fatty acids)
Slide25Biological Membranes Biological Membranes • the presence of cholesterol increases rigidity • the presence of cholesterol increases rigidity • with heat, membranes become more disordered; the transition temperature is higher for more rigid membranes; it is lower for less rigid membranes • with heat, membranes become more disordered; the transition temperature is higher for more rigid membranes; it is lower for less rigid membranes
Slide26Membrane Proteins Membrane Proteins • Functions: transport substances across membranes, receptor sites, and sites of enzyme catalysis • Functions: transport substances across membranes, receptor sites, and sites of enzyme catalysis • Peripheral proteins • Peripheral proteins • bound by electrostatic interactions • bound by electrostatic interactions • can be removed by raising the ionic strength • can be removed by raising the ionic strength • Integral proteins • Integral proteins • bound tightly to the interior of the membrane • bound tightly to the interior of the membrane • removed by treatment with detergents or ultrasonification • removed by treatment with detergents or ultrasonification • removal generally denatures them • removal generally denatures them
Slide27Fluid Mosaic Model Fluid Mosaic Model • Fluid: there is lateral motion of components in the membrane; • Fluid: there is lateral motion of components in the membrane; • proteins, for example, “float” in the membrane and can move along its plane • proteins, for example, “float” in the membrane and can move along its plane • Mosaic:components in the membrane exist side-by-side as separate entities • Mosaic:components in the membrane exist side-by-side as separate entities • the basic structure is that of a lipid bilayer with proteins, glycolipids, and steroids such as cholesterol embedded in it • the basic structure is that of a lipid bilayer with proteins, glycolipids, and steroids such as cholesterol embedded in it • no complexes, as for example, lipid-protein complexes, are formed • no complexes, as for example, lipid-protein complexes, are formed
Slide28Membrane Transport Membrane Transport • Passive transport • Passive transport • driven by a concentration gradient • driven by a concentration gradient • simple diffusion: a molecule or ion moves through an opening created by a channel protein • simple diffusion: a molecule or ion moves through an opening created by a channel protein • facilitated diffusion: molecule or ion is carried across a membrane by a carrier protein • facilitated diffusion: molecule or ion is carried across a membrane by a carrier protein • Active transport • Active transport • a molecule or ion is moved against a concentration gradient • a molecule or ion is moved against a concentration gradient • see the Na+/K+ ion pump • see the Na+/K+ ion pump
Slide29Membrane Receptors Membrane Receptors • Membrane receptors • Membrane receptors • generally oligomeric proteins • generally oligomeric proteins • binding of a biologically active substance to a receptor initiates an action within the cell • binding of a biologically active substance to a receptor initiates an action within the cell
Slide30Lipid-Soluble Vitamins Lipid-Soluble Vitamins • Vitamins are divided into two broad classes on the basis of their solubility • Vitamins are divided into two broad classes on the basis of their solubility . Those that are water-soluble . Those that are water-soluble . Those that are lipid-soluble (and hence classified as lipids) . Those that are lipid-soluble (and hence classified as lipids) The lipid-soluble vitamins include A, D, E, and K The lipid-soluble vitamins include A, D, E, and K
Slide31ProstaglandinsProstaglandins • Prostaglandins: a family of compounds that have the 20-carbon skeleton of prostanoic acid • Prostaglandins: a family of compounds that have the 20-carbon skeleton of prostanoic acid
Slide32ProstaglandinsProstaglandins • Prostaglandins are not stored in tissues as such, but are synthesized from membrane- bound 20-carbon polyunsaturated fatty acids in response to specific physiological triggers • Prostaglandins are not stored in tissues as such, but are synthesized from membrane- bound 20-carbon polyunsaturated fatty acids in response to specific physiological triggers • one such polyunsaturated fatty acid is arachidonic acid • one such polyunsaturated fatty acid is arachidonic acid
Slide33ProstaglandinsProstaglandins • among those synthesized from arachidonic acid are • among those synthesized from arachidonic acid are
Slide34ProstaglandinsProstaglandins • Research on the involvement of PGs in reproductive physiology has produced several clinically useful derivatives • Research on the involvement of PGs in reproductive physiology has produced several clinically useful derivatives • 15-Methyl-PGF2a is used as a therapeutic abortifacient • 15-Methyl-PGF2a is used as a therapeutic abortifacient
Slide35ProstaglandinsProstaglandins • the PGE1 analog, misoprostol, is used for prevention of ulceration associated with the use of aspirin-like non-steroidal antiinflammatory drugs (NSAIDs) • the PGE1 analog, misoprostol, is used for prevention of ulceration associated with the use of aspirin-like non-steroidal antiinflammatory drugs (NSAIDs)
Slide36LeukotrienesLeukotrienes • Leukotrienes: derived from arachidonic acid • Leukotrienes: derived from arachidonic acid • found in white blood cells (leukocytes) • found in white blood cells (leukocytes) • an important property is constriction of smooth muscles, especially in the lungs • an important property is constriction of smooth muscles, especially in the lungs
Slide37ThromboxanesThromboxanes • derived from arachidonic acid • derived from arachidonic acid • contain a four-membered cyclic ether within a six-membered ring induce platelet aggregation and smooth muscle contraction • contain a four-membered cyclic ether within a six-membered ring induce platelet aggregation and smooth muscle contraction