Energy Transformations and Matter States Vocabulary Test Review


In this minute to win it review, you will be quizzed on essential vocabulary related to energy transformations and matter states. You will be asked to identify the temperature at which a
- Uploaded on | 2 Views
-
 josephclark
josephclark
About Energy Transformations and Matter States Vocabulary Test Review
PowerPoint presentation about 'Energy Transformations and Matter States Vocabulary Test Review'. This presentation describes the topic on In this minute to win it review, you will be quizzed on essential vocabulary related to energy transformations and matter states. You will be asked to identify the temperature at which a. The key topics included in this slideshow are . Download this presentation absolutely free.
Presentation Transcript
Slide1Vocabulary& Essential Question Test Review
Slide2Minute to Win It10.1 Energy transformations 1. The temperature at which a substance turns into a solid 2. Energy of Motion 3. Matter that has no definite shape nor volume 4. To gain heat and change state from a solid to a liquid 5. Matter that has definite shape and volume 6. Stored energy caused by position or the shape of an object 7. Any form in which matter exists 8. Temperature at which a substance turns into a gas 9. Matter that has no definite shape but has definite volume 10. The ability to move and do work 11. To heat a liquid to a point where is changes to a gas 12. To lose heat and change state from a liquid to a solid. 13. Temperature at which a substance turns into a liquid. Freezing Point Kinetic Energy Gas Melt Solid Potential Energy State Boiling Point Liquid Energy Boil Freeze Melting Point
Slide3Review of Energy Transformationsand Conservation Multiple Transformations A series of energy transformations must occur for you to ride your bike. What are the forms of energy involved in each transformation? nuclear electromagnetic electromagnetic chemical chemical thermal chemical kinetic
Slide41.What does the Law of Conservation of Energy state? 2. Explain how this diagram demonstrates this Law Energy cannot be created or destroyed, it only changes form (transforms) In the diagram, energy transforms from chemical to heat and electrical, then finishes with thermal energy in the coffee. Energy was not created or destroyed, it just kept changing form.
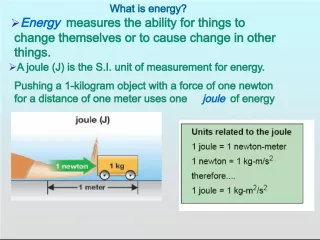
Slide5Minute to Win It10.1 Particle Energy & Law of Conservation 1. A type of potential energy that is stored in the nucleus of an atom 2. Potential energy stored in the bonds between the atoms of a substance 3. Total kinetic and potential energy of a substance; depends on its temperature and the number of particles 4. Energy of electrical charges 5. Form of energy that travels through space in waves; ex. Radio waves, light waves, and gamma rays. 6. Energy is neither created nor destroyed but changes form 7. A force between two objects that rub against each other Nuclear Energy Chemical Energy Thermal Energy Electrical Energy Electromagnetic Energy Law of Conservations of Energy Friction
Slide610.2 Thermal Energy, Temperature, & Heat1. Temperature scale used mostly in the U.S. 2. Total kinetic and potential energy of a substance; depends on its temperature and the number of particles 3. Temperature scale with water at 0=freezing and 100=boiling 4. Temperature scale with no negative numbers and is used by scientists 5. The average kinetic energy of the particles in an object (depends on speed of particles only) 6. No particle movement occurs at this point Farenheit Thermal Energy Celsius Kelvin Temperature Absolute Zero
Slide7Minute to Win It10.3 The transfer of Heat 1. The transfer of thermal energy from a warmer area to a cooler area. 2. Heat transfer that occurs when two objects are touching each other 3. The name of the circular pattern formed in this type of heat transfer 4. The transfer of heat by electromagnetic waves, does not require matter. 5. Type of heat transfer caused by changes in the density in fluids 6. Has no fixed shape and includes liquids and gases Heat Conduction Convection Current Radiation Convection Fluids
Slide8Essential Question Review1. Q: How does heat affect a system? 1. A: Heat can change temperature in a system and cause a change in state of matter 2. Q: Why can adding or removing heat from a system cause a change in state of matter? 2. A: It changes the speed of the particles moving in the substance (slow = cool; fast = hot) 3. Q: What are the 5 forces of Energy that are related to particles? 3. A: “TEEN-C” = Thermal, Electrical, Electromagnetic, Nuclear, and Chemical 4. Q: What does the Law of Conservation of Energy State? 4. A: Energy is neither created NOR destroyed, it only changes form 5. Q: What is Friction? 5. A: A force when two objects rub together; this creates heat
Slide9Essential Question Review6. Q: Explain how friction is a good example of energy being conserved 6. A: Friction shows energy is conserved (not created or destroyed) because two objects rubbing together transform kinetic energy into heat. 7. Q: What determines the temperature of an object? 7. A: The speed (how fast) the particles of are moving in the substance 8. Q: How is thermal energy different from temperature? 8. A: Thermal energy depends on the speed of the particles AND the number of particles, but temperature depends ONLY on the speed of the particles in the substance. 9. Q: How is heat transferred from one substance to another? 9. A: From the warmer substance to the cooler substance 10. Q: What are the 3 methods of heat transfer (describe each) 10. A: Conduction (objects touching), convection (density differences cause a circular pattern of fluids, radiation (EM waves heat things ex. Sun’s rays)
Slide10Self Check You may Check your work on IAN pages 94-96 to make sure your answers are right.
Slide11http://www.pbslearningmedia.org/asset/lsps07_int_heattransfer/Conduction Convection Radiation What will the molecules do? Use the website below to see how the molecules move in each type of heat transfer
Slide12Do IAN pg 94
Slide15conductionconve ction conve ction conve ction conduction 5. The cause of weather systems on Earth is _______________. conve ction
Slide16conductionconve ction conve ction conduction conduction conduction
Slide17Answer Review - IAN pg 96Question : How does an apple pie get cooked in an oven? Be sure to use the concepts of thermal energy, temperature, heat transfer, conduction, convection, and radiation to explain your answer. An apple pie gets cooked in the oven because of several types of heat transfer and an increase in the thermal energy of the apple pie. Turning on the oven causes a coil on the bottom of the oven to light up and heat the oven by radiation . The heat transfers from the hot coil to the colder oven and pie by radiation. As this heat transfers to the pie, the molecules vibrate faster, increasing the temperature and thermal energy of the pie. As the temperature rises, the inside of the pie cooks by convection because the water in the apples comes out as they cook. This provides a fluid inside the pie that will form a circular convection current due to differences in density that will make hot fluids rise, and cold fluids sink. The crust will continue to cook by radiation. This process may take about an hour. When it is done, you need to use potholders to take the hot apple pie out of the oven. If you don’t use potholders, you will probably burn your hands because the heat will transfer by conduction from the baking pan to your hands .