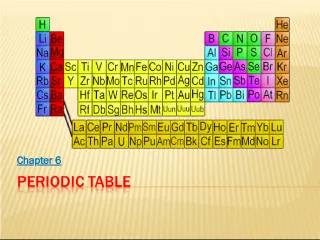
The Evolution of the Periodic Table


From Dmitri Mendeleev's discovery of a repeating pattern in 1869 to Henry Moseley's change in the arrangement in 1914, the periodic table has evolved. With gaps filled and predictions proven right, the periodic table remains a vital tool in chemistry.
- Uploaded on | 0 Views
-
 rachel88
rachel88
About The Evolution of the Periodic Table
PowerPoint presentation about 'The Evolution of the Periodic Table'. This presentation describes the topic on From Dmitri Mendeleev's discovery of a repeating pattern in 1869 to Henry Moseley's change in the arrangement in 1914, the periodic table has evolved. With gaps filled and predictions proven right, the periodic table remains a vital tool in chemistry.. The key topics included in this slideshow are Dmitri Mendeleev, periodic table, atomic mass, repeating pattern, atomic number,. Download this presentation absolutely free.
Presentation Transcript
2. Discovering a Pattern In 1869, Russian chemist Dmitri Mendeleev arranged the elements in order of increasing atomic mass. His Periodic table showed that Elements with similar properties occurred in a repeating pattern There were gaps in its pattern He could predict the properties of the missing elements By 1886, all of the gaps had been filled and Mendeleevs predictions were right.
3. Changing the Arrangement A few elements properties did not fit in the pattern of Mendeleevs table. 1914: British scientist Henry Moseley found the atomic number of atoms. Elements arranged by atomic number fit the pattern in Mendeleevs table.
4. Elements are arranged: Vertically into Groups Horizontally Into Periods
5. Why?
6. If you looked at one atom of every element in a group you would see
7. Each atom has the same number of electrons in its outermost shell. An example
8. The group 2 atoms all have 2 electrons in their outer shells Be (Beryllium) Atom Mg (Magnesium) Atom
9. The number of outer or valence electrons in an atom effects the way an atom bonds. The way an atom bonds determines many properties of the element. This is why elements within a group usually have similar properties.
10. If you looked at an atom from each element in a period you would see
11. Each atom has the same number of electron holding shells . An example
12. The period 4 atoms each have 4 electron containing shells K (Potassium) Atom Fe (Iron) Atom Kr (Krypton) Atom 4 th Shell
13. Each group has distinct properties The periodic Table is divided into several groups based on the properties of different atoms.
14. The Periodic Table and Classes of Elements Elements are classified as metals, nonmetals, and metalloids based on number of electrons in outer shell an outer shell is considered complete when it contains 8 electrons The zigzag line on the periodic table can help you recognize which elements belong in which category.
15. Group 1: Alkali Metals Alkali metals properties: metals 1 electron in the outer level very reactive softness, color of silver, shininess, low density
16. Alkali Metals Soft, silvery colored metals Very reactive!!! http://www.lyon. edu/webdata/U sers/DMcDowel l/GenChem/alka lishow.html http://www.lyon. edu/webdata/U sers/DMcDowel l/GenChem/alka lishow.html
17. found to the left of the zigzag line have few electrons in their outer energy level. shiny, ductile, malleable, and are good conductors of electric current and thermal energy. Metals
18. Alkali Metals reacting with water: Li (Lithium) Na (Sodium) K (Potassium) Rb (Rubidium) Cs (Cesium) What would you expect from Francium?!?!
19. Group 2: Alkaline-Earth Metals Alkaline-earth metals properties : metals 2 electrons in the outer level very reactive, but less reactive than alkali metals color of silver, higher densities than alkali metals
20. Alkaline Earth Metals Silvery-White Metals Fairly reactive Many are found in rocks in the earths crust
21. Group 312: Transition Metals Properties of Transition Metals vary widely but include: metals 1 or 2 electrons in the outer level less reactive than alkaline-earth metals shininess, good conductors of electric current and thermal energy
22. Transition Metals Malleable (easily bent/hammered into wires or sheets) Most are good Conductors of electricity
23. How many things can you think of that have Transition Metals in them?
25. Metalloids lie on either side of these stairsteps They share properties with both metals and non-metals Si (Silicon) and Ge (Germanium) are very important semi-conductors
26. border the zigzag line have about half of a complete set of electrons in their outer energy level. Metalloids have some properties of metals and some properties of nonmetals. Metalloids are also called semiconductors . Metalloids
27. What are semiconductors used in?
28. Nonmetals Brittle Do not conduct electricity
29. to the right of the zigzag line have an almost complete set of electrons in their outer energy level. not shiny, ductile, or malleable, and poor conductors of electric current and thermal energy. Nonmetals
30. Group 13: Boron Group Group 13 properties: one metalloid and five metals 3 electrons in the outer level reactive solids at room temperature
31. Group 14: Carbon Group Group 14 properties: one nonmetal, two metalloids, and two metals 4 electrons in the outer level reactivity varies among the elements solids at room temperature
32. Group 15: Nitrogen Group Group 15 properties: two nonmetals, two metalloids, and two metals 5 electrons in the outer level reactivity varies among the elements solids at room temperature (except for nitrogen, which is a gas)
33. Section 2 Grouping the Elements Group 16: Oxygen Group Group 16 properties: group contains three nonmetals, one metalloids, and one metal 6 electrons in the outer level reactive solids at room temperature (except for oxygen, which is a gas) Chapter 12
34. Section 2 Grouping the Elements Hydrogen The properties of hydrogen do not match the properties of any single group, so hydrogen is set apart. a nonmetal 1 electron in the outer level reactive colorless, odorless gas at room temperature, low density Chapter 12
35. Most are Poisonous Fairly reactive Halogens
36. Section 2 Grouping the Elements Group 17: Halogens Halogens are the elements in Group 17. Group 17 properties: group contains nonmetals 7 electrons in the outer level very reactive poor conductors of electric current, never in uncombined form in nature Chapter 12
37. Chlorine Gas was used as a chemical weapon during World War I. It was used by the Nazis in World War II.
38. Section 2 Grouping the Elements Group 18: Noble Gases Noble gases are the elements in Group 18. Group 18 properties: group contains nonmetals 8 electrons in the outer level (except helium, which has 2) unreactive colorless, odorless gases at room temperature Chapter 12
39. Unreactive Gases at room temperature Noble Gases
40. Jellyfish lamps made with noble gases artist- Eric Ehlenberger
41. Colors Noble Gases produce in lamp tubes: Ne (Neon) : orange-red Hg ( Mercury) : light blue Ar (Argon) : pale lavender He (Helium) : pale peach Kr (Krypton) : pale silver Xe (Xenon) : pale, deep blue
42. Group 312: Transition Metals, continued Lanthanides and Actinides Some transition metals from Periods 6 and 7 appear in two rows at the bottom of the periodic table. Elements in the first row are called lanthanides and elements in the second row are called actinides .
43. Lanthanide Series Actinide Series
44. The Periodic Tables Use the terms below to complete the concept map on the next slide. Chapter 12 Concept Map elements periods metals electrons nonmetals periodic table groups (families)
45. The Periodic Table Chapter 12
46. The Periodic Table Chapter 12
47. 1. Which of the following statements is correct for the elements shown? Chapter 12 Standardized Test Preparation A Lithium has the greatest atomic number. B Sodium has the least atomic mass. C Atomic number decreases as you move down the column. D Atomic mass increases as you move down the column.
48. 1. Which of the following statements is correct for the elements shown? Chapter 12 Standardized Test Preparation A Lithium has the greatest atomic number. B Sodium has the least atomic mass. C Atomic number decreases as you move down the column. D Atomic mass increases as you move down the column.
49. 2. Which of the following statements best describes the outer electrons in atoms of the elements shown? F The atoms of each element have 1 outer-level electron. G Lithium atoms have 3 outer-level electrons, sodium atoms have 11, and potassium atoms have 19. H Lithium atoms have 7 outer-level electrons, sodium atoms have 23, and potassium atoms have 39. I The atoms of each element have 11 outer-level electrons. Chapter 12 Standardized Test Preparation
50. 2. Which of the following statements best describes the outer electrons in atoms of the elements shown? F The atoms of each element have 1 outer-level electron. G Lithium atoms have 3 outer-level electrons, sodium atoms have 11, and potassium atoms have 19. H Lithium atoms have 7 outer-level electrons, sodium atoms have 23, and potassium atoms have 39. I The atoms of each element have 11 outer-level electrons. Chapter 12 Standardized Test Preparation
51. 3. The elements featured in the image belong to which of the following groups? A noble gases B alkaline-earth metals C halogens D alkali metals Chapter 12 Standardized Test Preparation
52. 3. The elements featured in the image belong to which of the following groups? A noble gases B alkaline-earth metals C halogens D alkali metals Chapter 12 Standardized Test Preparation
53. 2. A chemical company is preparing a shipment of 10 g each of four elements. Each element must be shipped in its own container that is completely filled with the element. Which container will be the largest? F the container of aluminum G the container of arsenic H the container of germanium I the container of silicon Chapter 12 Standardized Test Preparation Element Density (g/cm 3 ) Mass (g) Aluminum 2.702 10 Arsenic 5.727 10 Germanium 5.350 10 Silicon 2.420 10
54. 2. A chemical company is preparing a shipment of 10 g each of four elements. Each element must be shipped in its own container that is completely filled with the element. Which container will be the largest? F the container of aluminum G the container of arsenic H the container of germanium I the container of silicon Chapter 12 Standardized Test Preparation Element Density (g/cm 3 ) Mass (g) Aluminum 2.702 10 Arsenic 5.727 10 Germanium 5.350 10 Silicon 2.420 10